Microbiology Reference Laboratory: Identification of Pathogens
April 11, 2024
Learning Objective: Examine the reference laboratory process for the identification of pathogens.
After receiving the specimens collected in the POL, the microbiology laboratory promptly inoculates the appropriate culture media first and then prepares a smear of the specimen on a slide to be stained (FIGURE 35.11). This ensures that the swab is not contaminated by the slide. If two swabs are received from the same source, one may be used for the culture and the other to make the smear. The equipment and supplies in a microbiology laboratory vary with the size of the facility. Most laboratories have a refrigerator, an autoclave, a safety cabinet, a microscope, and an incubator.
Staining
Learning Objective: Discuss the identification of pathogens in the reference laboratory using various staining techniques.
Pathogenic microorganisms generally are colorless, and a microscope is needed to see them. Special stains (e.g., Gram stain and acid-fast stain) are used to differentiate bacteria based on cell membrane differences. As discussed previously, Gram stain differentiates bacteria into two categories according to the chemical makeup of the cell wall. The acid-fast stain differentiates bacteria into two categories based on the presence or absence of a waxy lipid in the cell wall.
Before staining can be done, the specimen must be applied to a labeled slide. The slide is then air-dried and fixed or adhered to the slide. Both heat (e.g., from a Bunsen burner or an incinerator) and methanol can be used to fix the sample to the slide. The heat and methanol cause protein in the sample to break down and stick to the slide. Note, that overheating the slide can cause cell distortion.
Gram Stain
The Gram stain, developed by Dr. Hans Christian Gram in the late 1800s, is still the most commonly used in microbiology. This procedure involves applying a sequence of reagents: a primary stain, mordant, decolorizer, and counterstain (secondary stain) to the slide. The dyes are taken up differently according to the chemical composition of the bacterial cell walls. Bacteria react best in the Gram stain when they are less than 24 hours old. Gram-positive bacteria stain purple, and gram-negative bacteria stain pink or red (see FIGURE 35.1). It is useful for the medical assistant to understand the procedures and the microscopic results obtained. For example, when a Gram stain report is called in, the terms GPCs and GNBs mean “gram-positive cocci” (deeply blue-stained circular cells) and “gram-negative bacilli” (pink/red stained rod-shaped cells).
35.7 Critical Thinking Application
What are the four reagents used in the Gram stain? What color are gram-positive organisms? What color are gram-negative organisms? Share your answers with the class.
Acid-Fast Stain
The acid-fast stain is used in the identification protocol for Mycobacterium species. M. tuberculosis causes tuberculosis and can be isolated from sputum or tissue samples. Mycobacterium avium complex (MAC) is a common soil organism that enters through the respiratory tract and spreads throughout the body. MAC is one of the causes of death among patients with AIDS. An overview of the acid-fast stain is listed here:
• A red primary dye, carbolfuchsin, is applied first
• Then the decolorizer, acid-alcohol, is applied
• Followed by a counterstain, methylene blue

FIGURE 35.11 Inoculating a blood culture plate with a swab.
Acid-fast positive microbes stain fuchsia red. Acid-fast negative microbes stain baby blue. Bacilli that are acid-fast positive often are referred to as acid-fast bacillus (AFB) (see FIGURE 35.2).
Inoculating Equipment
Learning Objective: Identify the inoculating equipment used by the reference laboratory in the process of identifying pathogens.
Next, the specimen must be spread on specific culture media based on the source of the specimen. The inoculated culture media are placed in a body temperature incubator to grow overnight. Inoculating needles and loops (FIGURE 35.12) are used to transfer samples to culture media or microbes to slides for staining. Needles and loops may be disposable and presterilized. They may also be made of wire and can be heat sterilized before and after each transfer (FIGURE 35.13). An inoculating loop is shaped like a bubble wand, and a thin film of liquid adheres to the loop. The amount of fluid held by the loop can be calibrated. For example, a urine culture uses a loop that delivers a 1-mcL sample. The urine in the loop is spread across the culture medium and allowed to grow overnight. The next day, each bacterium becomes a visible colony. Colonies can then be counted and analyzed to determine the cause of a urinary tract infection.

FIGURE 35.12 Inoculating needles (left). Inoculating loops (right). Courtesy Simport Plastics, Beloeil, Quebec, Canada.

FIGURE 35.13 Incinerator for sterilizing wire loops and needles.

FIGURE 35.14 Positive strep test result on blood agar (left side of plate). Group A streptococcus (GAS) shows beta hemolysis (clearing) of the blood agar below the growing colonies. Notice there is no hemolysis or clearing of the blood around the white bacitracin disk. That is because bacitracin can destroy GAS.
Assessing a Culture
Learning Objective: Describe the reference laboratory assessment of a throat culture and urine culture.
When the original (primary) culture has incubated at the appropriate temperature for 18 to 24 hours, it is examined for evidence of pathogens. Because normal flora is often present in samples in addition to pathogens, a trained eye is required to spot the organisms that might be causing an infection. Suspicious colonies are subcultured onto the appropriate medium to isolate them in pure culture. When the organism is in pure culture, staining and additional biochemical testing can be done to identify the organism. Throat and urine cultures may be performed in POLs that have been CLIA certified to perform moderately complex testing.
Throat Cultures
Streptococcus pyogenes, also known as group A strep (GAS) or beta-hemolytic streptococcus, causes strep throat. If not diagnosed and treated promptly, this infection can cause severe complications, including scarlet fever, rheumatic fever, and glomerulonephritis. A throat swab is collected from the patient’s throat. Then the swab is streaked for isolation on a 5% sheep’s blood agar plate (BAP) . An antibiotic disk is placed on the first quadrant of the streaked plate (FIGURE 35.14) and incubated overnight at 37°C (98.6°F). The antibiotic disk contains bacitracin, which prevents the growth of S. pyogenes. Complete clearing of the agar around the colonies indicates beta hemolysis, which is caused by a toxin produced by S. pyogenes. The toxin breaks down the red blood cells in the agar, causing the agar to be a clear golden color around the colonies. The presence of beta hemolysis and a zone of no growth around the disk indicate that the patient has strep throat (see FIGURE 35.14). Additional testing may be needed to confirm the identity of the organism.
Streaking for Isolation
Streaking for isolation is also called a four-quadrant streak. It is a procedure that is performed with most microbiology specimens. By distributing bacteria across the agar plate and isolating bacterial colonies, laboratory personnel can see characteristics of the bacteria that are useful in the identification process.
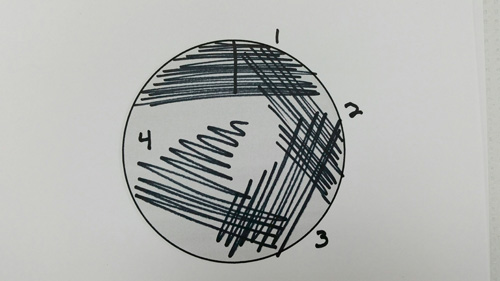
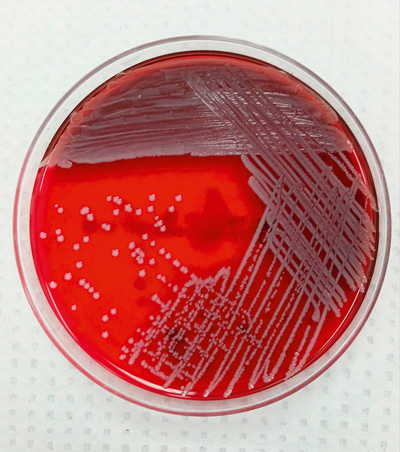
Urine Cultures
With urine cultures, the bacterial colonies that appear after incubation are counted. A calibrated inoculating loop is dipped into a well-mixed urine sample that was collected by the clean-catch midstream (CCMS) method or by catheterization. The urine from the loop is spread on solid culture media and incubated for 18 to 24 hours at 37°C (98.6°F). Each colony that grows on the plate represents 1000 colony-forming units (cfu) per milliliter. The final cfu results are interpreted as follows:
• Normal: <10,000 cfu/mL of urine; no urinary tract infection (UTI) is present
• Borderline: 10,000 to 100,000 cfu/mL of urine; a chronic or relapsing infection may be present, and the test should be repeated
• Positive: >100,000 cfu/mL of urine; a UTI is likely
The medical assistant needs to be aware of the terminology and values reported for urine cultures.
Microbiology Culture and Sensitivity Testing
Learning Objective: Explain culture and sensitivity testing.
Once a bacterial infection has been identified, additional steps are required for successful treatment. To determine the appropriate antibiotic to destroy the pathogen, the provider will order a culture and sensitivity (C&S) test. Culture refers to growing the organisms, and sensitivity refers to the organism’s susceptibility to antibiotics.
The healthcare provider must decide which medication to order based on initial test results and the patient’s physical examination. C&S test results provide vital information about which specific antibiotics work best against the particular infective pathogen.
There are many methods of performing sensitivity testing. Some methods involve culture media and antibiotic disks. Other methods are fully automated and require a very small amount of the pure culture to be tested. No matter what method is used, sensitivity testing is reported to the provider in one of three categories for each antibiotic tested:
• S means that the pathogen is susceptible, or that the antibiotic is effective in destroying that particular organism.
• R means that the pathogen is resistant, or that the antibiotic is not effective in destroying that particular organism.
• I means intermediate, or that additional testing must be performed to determine the dosage of antibiotic necessary for successful treatment.
Some testing methods may give additional information regarding the dosage tested in vitro . This information may be helpful as the providers choose the appropriate antibiotic for treatment. The appropriate antibiotic agent meets the following criteria:
• Destroys the infectious agent with a reasonable level of the drug
• Is the least toxic to the patient and has the least impact on normal flora of the body
• Has the desired pharmacologic characteristics (preparation, route of delivery, effectiveness)
• Is the most economical
35.8 Critical Thinking Application
Laura is looking over the reference laboratory results from the day. Mrs. Liz Darcy was in for an office visit yesterday and collected a urine specimen for C&S. Her preliminary culture report came back as 82,000 cfu/mL, pure culture. The identification and susceptibility report is due to follow tomorrow.
• Does Mrs. Darcy have a urinary tract infection?
• Does Mrs. Darcy need any additional testing for this condition?
• In your own words, define pure culture.
• Describe C&S in your own words.
